
In my lab we focus on the design of innovative drug-delivery systems and employing advanced mass spectrometry imaging technologies to address fundamental questions in medicine. We believe that mass spectrometry imaging can not only elucidate mechanistic pathways of diseases, but can also identify novel therapeutic targets and guide us in rational design of drug delivery systems.
Mass spectrometry imaging has emerged in recent years as a powerful tool for directly measuring the distribution of molecules in tissue sections. The uniqueness of this technique is that it enables a high-resolution outlook on how various substances distribute in organs at different time points without prior knowledge of their existence or employing fluorescent or radioactive labels. Hundreds of molecular species can be visualized within one single step, which can reflect on the subtlest physiological processes triggered by pathological conditions or external intervention, such as drug delivery.
I am a pharmacist and a chemist by training. My academic journey began as an undergraduate at the Hebrew University School of Pharmacy, and continued at Casali Institute of Applied Chemistry, the Hebrew University, where I did my graduate studies. My PhD thesis centered on designing drug delivery systems for poorly water-soluble pharmaceutics. I developed a general method allowing a rapid conversion of almost any insoluble substance into a dry power composed of easily water-dispersible nanoparticles. In my postdoctoral research at Stanford University I employed mass spectrometry imaging to study a variety physiological processes in living tissues, to distinguish between tissues of various pathologies, and to guide drug discovery. This work convinced me that mass spectrometry imaging can provide answers to many essential questions in modern medicine and pharmaceutics.
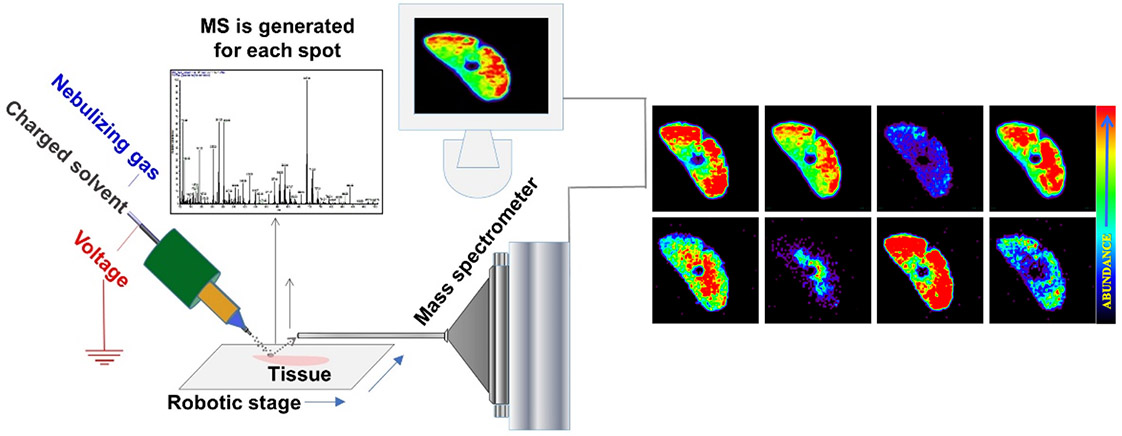
Fig.1. Schematic presentation of ambient mass spectrometry imaging technique - Desorption Electrospray Ionization Mass Spectrometry Imaging.
A beam of charged droplets is directed onto a tissue surface to desorb and ionize molecules, and the splash of these droplets carries the resultant ions into a mass spectrometer for analysis. A two-dimensional imaging stage moves the tissue at a controlled speed to record the mass spectra from different spatial coordinates and the signal is subsequently converted into images of molecular ion distributions. Each spectrum generates a single pixel in the image, whereas every mass peak can be translated into a separate color-coded map based on its intensity and the spatial distribution of its corresponding molecular ion.

