The primary focus of the research in my laboratory is the regulation of ion transport across the plasma membrane of eukaryotic cells. In particular, we study the main transport system for sodium and potassium, the sodium-potassium-ATPase, and its regulation by cardiac steroids.
Identification of endogenous cardiac steroids in mammalian tissue; The biological consequences of the interaction of cardiac steroids with the sodium-potassium-ATPase; Biosynthesis of the cardiac steroids in the adrenal gland; Effects of endogenous sodium-potassium-ATPase inhibitors on cell differentiation; Determination of the levels of endogenous sodium-potassium-ATPase inhibitors in pathological states, including hypertension, preeclampsia; malignancies (cancer) and manic depressive illnesses; Involvement of the sodium-potassium--ATPase/cardiac steroids system in depressive disorders; Involvement of the sodium-potassium-ATPase/cardiac steroids system in cardiac function; Involvement of intestinal signals in the regulation of phosphate homeostasis; Volume regulation and its involvement in the mitogenic response.
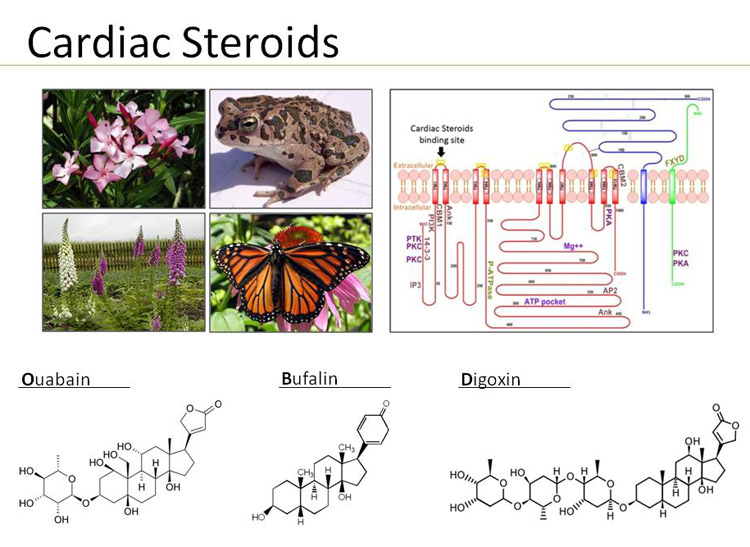
Cardiac steroids, such as ouabain, digoxin and bufalin are hormones synthesized by and released from the adrenal gland and the hypothalamus. These compounds, the structure of which resembles that of plant and amphibian and butterfly steroids, interact only with the plasma membrane Na+, K+-ATPase (Figure 1). This interaction elicits numerous specific biological responses affecting the function of cells and organs.
Topics Currently under investigation include
Involvement of the sodium-potassium--ATPase/cardiac steroids system in depressive disorders
Depressive disorders, including major depression, dysthymia and bipolar disorder, are a serious and devastating group of diseases that have a major impact on the patients’ quality of life, and pose a significant concern for public health. The etiology of depressive disorders remains unclear. The Monoaminergic Hypothesis, suggesting that alterations in monoamine metabolism in the brain are responsible for the etiology of depressive disorders, is now recognized as insufficient to explain by itself the complex etiology of these diseases. Data from our and other laboratories has provided initial evidence that endogenous cardiac steroids and their only established receptor, the Na+, K+-ATPase, are involved in the mechanism underlining depressive disorders, and BD in particular.
Our recent studies in this project (Eur. Neuropsychopharmacol. 22:72-729, 2012; Front Endocrinol (Lausanne). 5:20, 1-13, 2014; Bipolar Disorders. 18:451-459, 2016; Brain Res. Bull. 137:356-362, 2018) showed that drugs affecting the Na+, K+-ATPase/cardiac steroids system are beneficial for the treatment of depression and that protection from oxidative stress is involved in this phenomenon. Hence our work is in accordance to the proposition that mal functioning of the Na+, K+-ATPase/cardiac steroids system may be involved in manifestation of depressive disorders and identify new compounds as potential drug for the treatment of these maladies.
Involvement of the sodium-potassium-ATPase/cardiac steroids system in cardiac function
The classical and best documented effect of cardiac steroids, as their name implies, is to increase the force of contraction of heart muscle. Indeed, cardiac steroids were widely used in Western and Eastern clinical practices for the treatment of heart failure and atrial fibrillation. Despite extensive research, the mechanism underlying cardiac steroids actions have not been fully elucidated. The dogmatic explanation for cardiac steroids-induced increase in heart contractility is that the inhibition of Na+, K+-ATPase by the steroids causes an increase in intracellular Na+ which, in turn, attenuates the Na+/Ca++ exchange, resulting in an increased intracellular Ca++ concentration, and hence greater contractility. However, recent observations led to the hypothesis that the ability of cardiac steroids to modulate a number of intracellular signaling processes, including that of extracellular-signal-regulated kinase (ERK) and PI3K-Protein Kinase B (AKT) pathways may be responsible for both short- and long-term changes in CS action on cardiac function. In recent studies on zebrafish (J Pharmacol Exp. Ther. 357:345-56, 2016) and rat (J. Cardiovasc. Pharmacol. Ther. 24: 78-89, 2019) experimental models, we proved this concept by demonstrating that the addition of ERK inhibitors (in-vitro and in-vivo) attenuated the CS-increase in heart contractility. Surprisingly, we have discovered that the combined treatment with CS and AKT inhibitors has beneficial effect as compared to the treatments with CS alone (J. Cardiovasc. Pharmacol. Ther. 24: 78-89, 2019). Treatment of hearts, in-vivo (or cardiomyocytes and isolated heart, in-vitro) resulted in a positive inotropic effect of the CS at doses that did not have any effects when administered alone (sub-nanomolar). This beneficial effect of the combined treatment with CS and AKT inhibitors was now proven in several experimental systems including, primary cardiomyocytes from zebrafish heart, zebrafish heart contractility in-vivo, primary cardiomyocytes from rat heart, isolated rat heart contractility (Langendorff preparation) and experiments on heart contractility following LAD ligation in rats. Hence, taken together, these studies establish a strong proof-of-principal for the development of the combined treatment for clinical use for the treatment of heart failure.

