Research Interests
We are interested in understanding how the process of alternative RNA splicing (a fundamental step of gene expression) is de-regulated in cancer and other genetic diseases, and how the proteins that control this process (splicing factors) contribute to cancer development and progression. We use both in vitro and in vivo cellular and animal model systems to identify the role of splicing factors in human diseases. A major effort in the lab is to translate our molecular findings into therapy. Based on the knowledge we have gathered, identifying oncogenic splicing factors and cancer-promoting splicing events that drive cancer development and progression, we are developing methods to intervene in this process. We have developed decoy oligonucleotides, as well as small molecules, to modulate the activity of oncogenic splicing factors and splice-switching antisense oligonucleotides to modulate oncogenic splicing events. An additional research direction aimed to inhibit cancer involves the generation of neoantigens to enhance immunotherapy by splicing modulation.
A new research effort in the lab focuses on understanding the regulation of mRNA stability by m6A RNA modifications and using its modulation to stabilize mutant genes for the treatment of both genetic diseases, such as Duchenne Muscular Dystrophy, as well as specific cancers.
Several of these applications have been patented and are being commercialized by the technology transfer company of the Hebrew University, Yissum. During 2021, three biotech companies were founded based on some of our RNA therapeutics technologies: SKIP therapeutics, Andlit Therapeutics and RNAble.
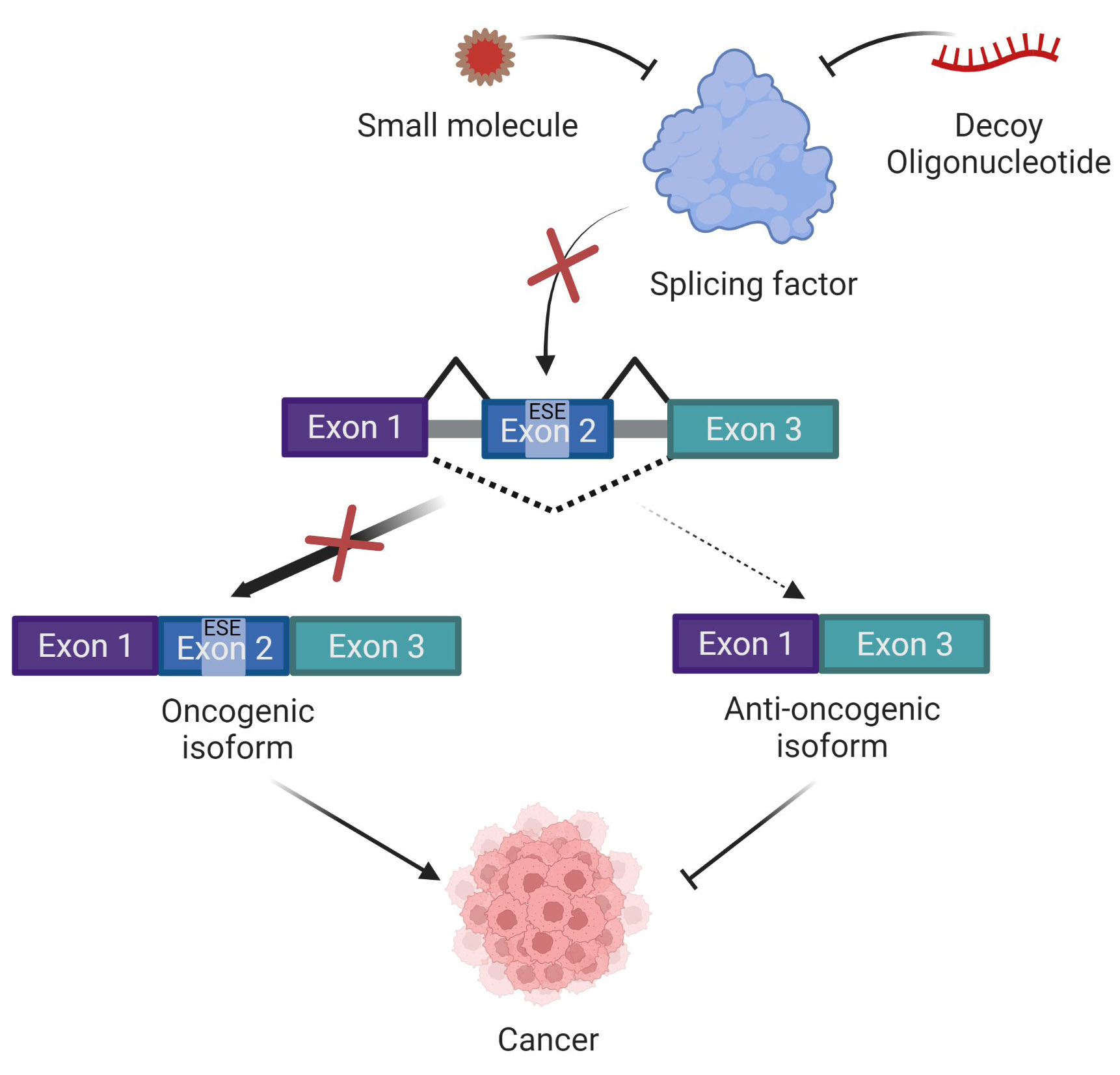 Targeting splicing factors using decoy oligonucleotides or small molecules.
Targeting splicing factors using decoy oligonucleotides or small molecules.
Dysregulated/mutated splicing factors can bind to a specific motif within the pre-mRNA and induce inclusion/skipping of exons, leading to the generation of tumor promoting oncogenic isoforms. Small molecules or decoy oligonucleotides can be used to inhibit the activity of the splicing factor, shifting the balance towards the generation of anti-oncogenic isoforms, thus inhibiting cancer.

